Вести
- 28.07 2023Клиниката за гастроентерохепатологија се вклучи во активностите кои во соработка со здружението “Хепар Центар” - Битола се организираат по повод одбележување на 28 Јули - Светскиот ден за борба против хепатитот.
- 12.07 2023Клиниката за гастроентерохепатологија стана член на Конзорциумот на Европската асоцијација за проучување на црниот дроб (EASL) и Европската фондација за проучување на хронична црнодробна слабост (EFCLIF) чии активности се насочени кон продлабочување на научните истражувања за хронична црнодробна слабост.
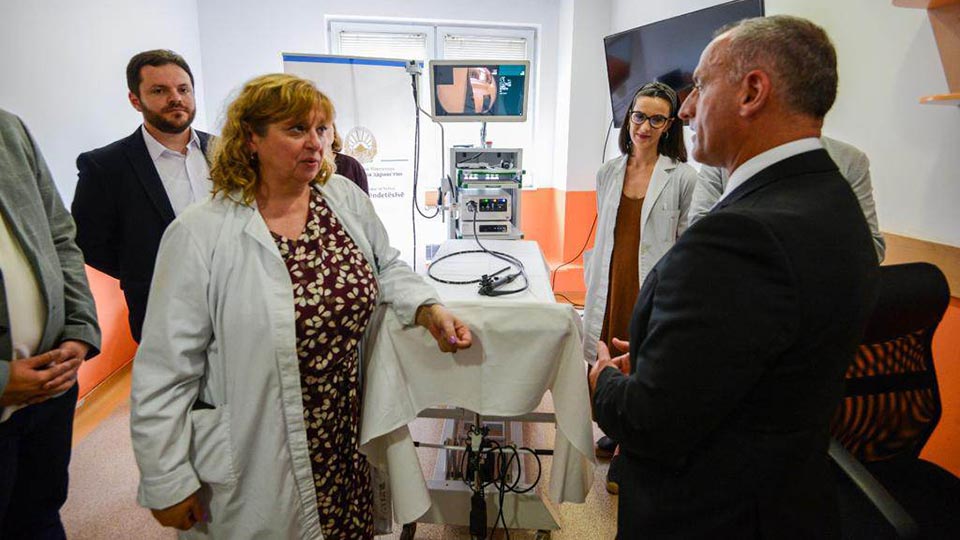
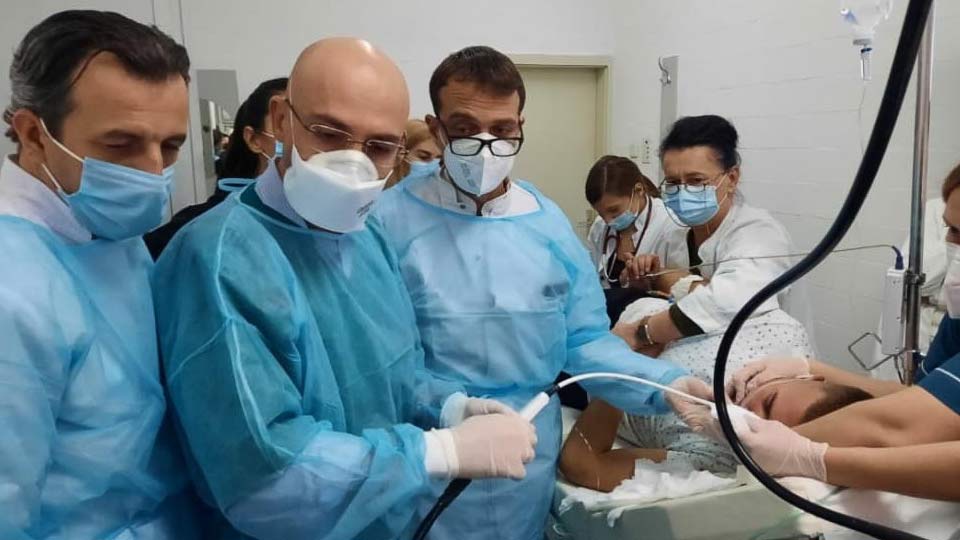
- 06.06 2023Во рамки на Клиниката за гастроентерохепатологија беше пуштен во функција нов Центар за болести на црн дроб кој е опремен со најсовремени апарати за дијагностика и третман. Проф. д-р Калина Гривчева Старделова, директорка на Клиниката, појасни дека овој центар е составен од повеќе делови и се осврна на придобивките за пациентите.

test komp 1
ЈЗУ УК Гастроентерохепатологија Скопје претставува установа со долготрајна традиција и искуство во давање и вршење на здравствени услуги, научно-истражувачка и образовна дејност, со целосна посветеност кон потребите и задоволството на пациентите.
Соопштенија
- 07.11 2023Посета на пациенти е дозволена само во времето предвидено за посета. Времето за посета е истакнато на влезните врати на одделите каде лежат пациентите. Воколку постои зголемен ризик од пренос на инфекции може да постои забрана за посета на пациенти. Забраната треба да биде истакната на влезната врата од одделот.
- 28.07 2023Клиниката за гастроентерохепатологија се вклучи во активностите кои во соработка со здружението “Хепар Центар” - Битола се организираат по повод одбележување на 28 Јули - Светскиот ден за борба против хепатитот.
- 06.06 2023Во рамки на Клиниката за гастроентерохепатологија беше пуштен во функција нов Центар за болести на црн дроб кој е опремен со најсовремени апарати за дијагностика и третман. Проф. д-р Калина Гривчева Старделова, директорка на Клиниката, појасни дека овој центар е составен од повеќе делови и се осврна на придобивките за пациентите.
Правилник за посета на лежечки пациенти
07.11.2023
Посета на пациенти е дозволена само во времето предвидено за посета. Времето за посета е истакнато на влезните врати на одделите каде лежат пациентите.
Воколку постои зголемен ризик од пренос на инфекции може да постои забрана за посета на пациенти. Забраната треба да биде истакната на влезната врата од одделот.
Заврши реновирањето на Клиниката за гастроентерохепатологија
24.03.2023
Денеска на Клиниката за гастроентерохепатологија се одбележа завршување на реновирањето на првиот кат, што практично опфаќа половина од просторот со кој располага оваа здравствена установа. Со проектот за реконструкција комплетно се обновени одделите за интензивна и полуинтензивна нега, каде се лекуваат пациентите во најкомплексна здравствена состојба, кабинетите за дијагностички процедури, работните простории за здравствениот персонал.
Во функција нов центар за болести на црниот дроб
06.06.2023
Во рамки на Клиниката за гастроентерохепатологија беше пуштен во функција нов Центар за болести на црн дроб кој е опремен со најсовремени апарати за дијагностика и третман. Проф. д-р Калина Гривчева Старделова, директорка на Клиниката, појасни дека овој центар е составен од повеќе делови и се осврна на придобивките за пациентите.
Клиниката за гастроентерохепатологија доби Сертификат за членство во EASL - CLIF Конзорциум
12.07.2023
Клиниката за гастроентерохепатологија стана член на Конзорциумот на Европската асоцијација за проучување на црниот дроб (EASL) и Европската фондација за проучување на хронична црнодробна слабост (EFCLIF) чии активности се насочени кон продлабочување на научните истражувања за хронична црнодробна слабост.